
However, greater than 85% of all ADA-deficient patients are infants with SCID. For example, a clinically related immunodeficiency occurs as a consequence of defects in the purine nucleotide salvage enzyme, purine nucleoside phosphorylase (PNP). Not all forms of SCID are caused by defects in ADA. In addition, S-adenosylhomocysteine hydrolase activity is markedly inhibited by 2′-deoxyadenosine resulting in accumulation of S-adenosylhomocysteine which in turn results in reduced synthesis of S-adenosylmethionine (AdoMet), a critical substrate in transmethylation reactions. The inhibition of RR leads to a block to DNA replication which is critical for the expansion of lymphocytes in response to an antigenic challenge.ĭeoxy-ATP also induces DNA strand breakage in non-dividing lymphocytes via a direct activation of a major protease (caspase 9) involved in apoptosis (programmed cell death).  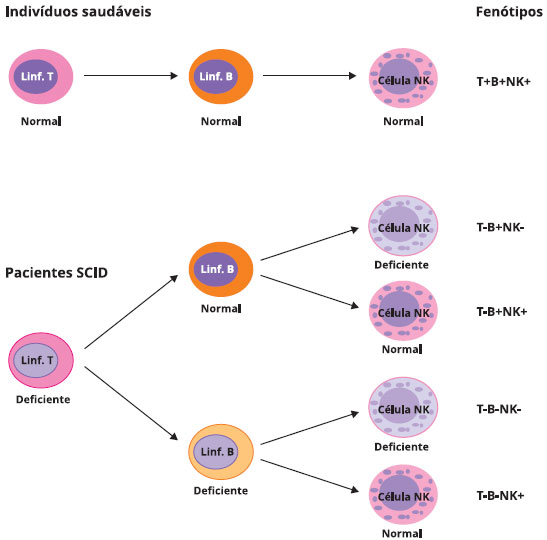
The consequences of increased dATP pools is an inhibition of ribonucleotide reductase (RR), the enzyme responsible for generating deoxyribonucleotides (necessary for DNA replication) from ribonucleotides. Increases in 2′-deoxyadenosine, through the action of ubiquitous nucleoside phosphorylases, results in dramatic increases in cellular dATP pools. The consequences of the elevations in these two ADA substrates are impaired lymphocyte differentiation, function, and viability which results in lymphopenia and severe immunodeficiency. In ADA deficiency there is an elevation in the level of adenosine and 2′-deoxyadenosine in the blood and 2′-deoxyadenosine levels in the urine are also elevated. Biochemical Consequences of ADA Deficiency Nearly 70% of all ADA mutations result in single amino acid substitutions with the rest being deletions and splicing mutations. Over 60 different mutations have been identified in the ADA gene resulting in immunodeficiency. These mRNAs encode proteins of 363 amino acids (isoform 1), 228 amino acids (isoform 2), and 339 amino acids (isoform 3). The ADA gene is located on chromosome 20q13.12 and is composed of 12 exons that generate three alternatively spliced mRNAs. Molecular Biology of ADA Deficient SCIDĪdenosine deaminase is encoded by the ADA gene. The level of expression of ADA also decreases as B cells mature. In addition to the differences in levels of ADA in various tissues, the enzyme is also developmentally regulated such that immature thymocytes express higher levels than do mature thymocytes. Reaction catalyzed by adenosine deaminase (ADA)Īlthough ADA activity is found in all tissues of the body, the highest concentrations are found in the thymus and other lymphoid tissues. Deficiency in purine nucleotide phosphorylase (PNP), another purine catabolic enzyme, results in 1%-2% of all cases of SCID.ĪDA catalyzes the irreversible deamination of adenosine and deoxyadenosine to inosine and 2′-deoxyinosine, respectively. This form of SCID is inherited as an autosomal recessive disorder with an incidence rate as high as 1:200,000, although some studies report an incidence of 1:1,000,000. The second most common (15%) form of SCID is caused by defects in the activity of the purine nucleotide catabolism enzyme, adenosine deaminase (ADA). The most common (45%) cause of SCID is the X-linked recessive disorder resulting from loss of function of the common gamma (γ) chain of the T-cell receptor and other interleukin (IL) receptors. There are at least 13 known and characterized genetic causes of SCID. Severe combined immunodeficiency (SCID) refers to a group of potentially fatal disorders due to a combined loss of function of both T- and B-lymphocytes. Clinical Features of ADA Deficient SCID.Biochemical Consequences of ADA Deficiency.Molecular Biology of ADA Deficient SCID.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
